K3iii: Opioid Syllabus – Opioids
Definitions
- Alkaloid = nitrogen containing base of a plant origin
- Opium = dry, powdered mixture of alkaloids obtained from poppy plant
- The principle alkaloid (50%) is M but there are many alkaloids in the mixture
- Opiate = a drug derived from opium
- Opioid = all exogenous substances that bind to opioid receptors to produce some agonist effect
- Endogenous opioid = family of endogenous opioid peptide ligands which act by binding opioid receptors (endorphins, dynorphins, enkephalins)
- Narcotic = Gk = stupor, any compound producing ‘numbness’ to external stimuli
Classification
- ORIGIN
- CHEMICAL
- CLINICAL PROFILE
Origin
- Exogenous/endogenous → endorphins, dynorphins, enkephalins
Chemical
- Natural alkaloid
- PHENANTHRENES → morphine, codeine
- Semi-synthetic
- Preserved fine ring skeleton (not essential for opioid activity)
- But alterations to major functional groups → heroin, oxycodone, buprenorphine
- Synthetic
- Fine ring structure is altered
- Do not have structure of Morphine, but contain phenanthrene nucleus of morphine
- Not a chemically modified version of M, they are manufactured by denovo synthesis
PHENYLPIPERIDINES → PETHIDINE, FENTANYL, REMI, ALFENTANYL
Clinical Profile
- Latency (time to peak) & duration
- Ultrashort latency + ultrashort DoA → REMIFENTANYL
- Ultrashort latency + short DoA → ALFENTANYL
- Short latency, intermediate DoA → FENTANYL, PETHIDINE
- Intermediate latency, long DoA → MORPHINE
- Specificity & efficacy
- Agonist
- Partial agonist
- Antagonist
Properties
- CEUTIC all opioids are H2O soluble salt preparations
Physical Properties
Drugs
pKa
M
7.9
Pethidine
8.5
F
8.4
Alfentanyl
6.5
REMI
7.1
Methadone
9.3
Drugs
% unionised
M
23%
Pethidine
5%
F
10%
Alfentanyl
90%
REMI
67%
Methadone
1%
Drugs
n-octanol/H2O
M
1.4
Pethidine
39
F
860
Alfentanyl
130
REMI
17.9
Methadone
115
Drugs
VD CC (L/kg)
M
0.2
Pethidine
1.5
F
0.7
Alfentanyl
0.2
REMI
0.07
Methadone
–
Drugs
VD SS (L/kg)
M
3.2
Pethidine
9.4
F
4
Alfentanyl
0.86
REMI
0.4
Methadone
6
Drugs
PPB
M
35%
Pethidine
65%
F
85%
Alfentanyl
90%
REMI
80%
Methadone
85%
Drugs
Clearance
(mL/kg/min)
M
15
Pethidine
10
F
13
Alfentanyl
6.4
REMI
50
Methadone
1.5
Drugs
HER
M
0.7
Pethidine
0.8
F
0.9
Alfentanyl
0.4
REMI
N/A
Methadone
–
Drugs
Dist t ½ (min)
M
15
Pethidine
10
F
13
Alfentanyl
11.6
REMI
1 min
Methadone
0.9
Drugs
Elim t ½ (hrs)
M
3
Pethidine
3 – 5
F
3.6
Alfentanyl
1.6
REMI
5 min
Methadone
35
Drugs
t½ keo (min)
M
Pethidine
F
4 – 5mins
Alfentanyl
0.6 – 2.3
REMI
1.5 min
Methadone
Drugs
C5HT (after 4hr infusion)
M
Pethidine
F
4hrs
Alfentanyl
60 mins
REMI
4 min
Methadone
- Ionisation = ↓lipid solubility
- Low octanol/H2O coefficient = hydrophilic
- Morphine = poor lipid solubility → because -OH at C3 & C6
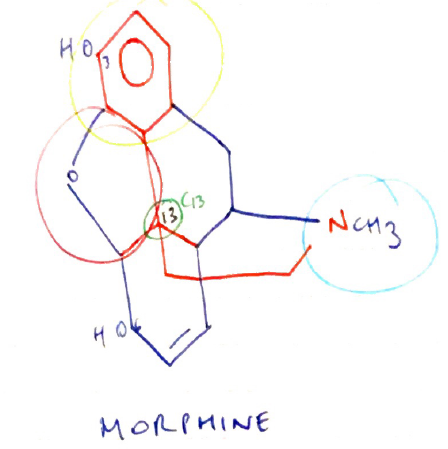
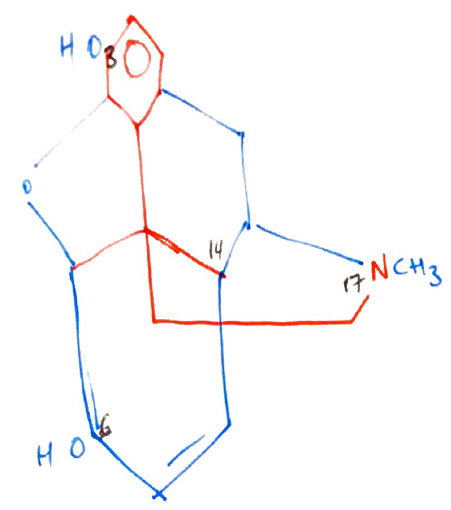
Structure
- Amine group = essential for opioid agonist activity
- +ve charge necessary to bind strongly to ANIONIC opioid receptor
- Aromatic ring = phenol group attached to quaternary carbon
- Quaternary carbon (C13) = carbon atom attached to four others → it is chiral & allows existence of enantiomers → attached to Phenolic Ring & Ethane Chain
- Ethane chain ( – CH2 – CH2 – ) = separates quaternary carbons from N atom in amine group
Isomerism
- Dextro & Levo enantiomers possible due to CHIRAL CARBON (C13)
- Only levoratory exhibits agonist activity
SAR
- C3 = maximum opioid potency
- Methylating this group (adding CH3) = makes codeine
- Codeine x 10 less potent cf. M
But Codeine has reduced Metabolism → ∴ codeine has better OBA cf. M
- C6 = addition of large side chains to C6 = ↑POTENCY because ↑lipid soluble e. HEROIN has a larger side chain at C6 (but this potency partially offset because also has masked the -OH at C3)
- N17 = Substitution of phenylethyl group into nitrogen17 ↑agonist activity i.e. FENTANYL, as well as ↑lipid solubility
- C14 = replacing nitrogen methyl group at N17 is coupled to C14 hydroxylation = formation of PURE ANTAGONIST → Naloxone
Indications, Administration, Dosing
Indications
- Analgesia
- Premed
- Attenuate CV response to laryngoscopy
- As a primary anaesthetic agent
- Tx heroin withdrawal
Route
- Enteral
- Oral → M, Codeine, Endone, Tramadol, Methadone
- PR → Oxycodone
- Parenteral
- SC, IM, IV → M, F, Pethidine
- Neuraxial → M, F, Pethidine
- Epidural = 10% IV dose
- Intrathecal = 10% epidural dose
- Topical
- F, Buprenorphine
Dose → google ANZCA opioid dose equivalent for their PDF statement
Opioid
Morphine
Oral
30mg
Parenteral
10mg
Codeine
200mg
120mg
Endone
15mg
15mg
Pethidine
240mg
100mg
Fentanyl
–
100mcg
Alfentanil
–
1mg
Remifentanil
–
100mcg
Mechanism of Action
- Opioids produce effect by binding µ opioid receptor
- µ opioid receptor located throughout CNS, but concentrated in periaqueductal grey matter near 4th ventricle & substantia gelatinosa of dorsal horns of SC
- Gi GPCR
- Closure of voltage sensitive Ca2+ channels on presynaptic membrane → ↓intrac Ca2+
- Post-synaptic stimulation of K-efflux → hyperpolarisation
- Inhibition of AC → ↓cAMP
- OVERALL → ↓cell membrane excitability, ↓transmission of nociceptive signals
Opioid Receptors
M receptor
2 subtypes
- µ1 = supraspinal analgesia
- Analgesia
- Miosis
- Euphoria
- µ2 = spinal analgesia
- Resp D
- Inhibits GI motility
- Euphoria
- Dependence
- Urinary retention
- Pruritus
Prototype agonist = MORPHINE
δ-receptor
2 subtypes
- Activation results in analgesia & resp depression
- Spinal analgesia
Prototype agonist = ENKEPHALINS
k-receptor
3 subtypes
- Activation results in:
- Miosis
- Dysphoria
- Inhibition ADH release (diuresis)
- Less analgesia
- Dependence
Prototype against = DYNORPHINS
Opioid Receptor Activation
- Endogenous peptide opioids (Endorphins, dynorphins, enkephalins) activate their specific opioid receptor
- Overall ↓ neurotransmission
- PRESYNAPTIC INHIBITION of NT release (Ach, DA, NA, sub P)
- POST-SYNAPTIC INHIBITION of NT release
- Opioid drugs mimic the actions of endogenous ligands
- The effect the drug produces depends on:
- Affinity for opioid receptor
- +ve charged amine essential to bind anionic opioid R
- ↑opioid receptor occupancy = ↑opioid effects
- Specificity of binding: µ, k, δ receptor
- Efficacy of opioid
- Affinity for opioid receptor
- Full agonist = maximal biological response
- Partial agonist = submaximal response
- Antagonist = no biological effect
- Mixed agonist-antagonist = simultaneously an agonist one receptor & an antagonist at another
Affinity & Efficacy of Opioids at Different Receptors
Full Agonist
Morphine
µ
+++
k
+
σ
Pethidine
+++
+
Fentanyl
+++
+
Alfentanil
+++
+
Remifentanil
+++
+
Partial Agonist
Buprenorphine
Partial Agonist
Nil
Tramadol
Partial Agonist
Nil
Antagonist
Naloxone
−−−
−
Pharmacodynamic Effects
CNS
- Analgesia
- Mainly due to peripheral opioid rec activation
- ↑pain threshold (↑ stimulus required to activate nociception)
- ∆ pain perception/response → Europhia
- Sedation
- Dull C fibres > Sharp Aδ fibres
- Sedation
- Miosis
- ↑parasymp outflow of CNS
Resp
- Ventilatory depression
- ↓CO2 responsiveness
- Dose dependent respiratory depression
- µ receptor of ventrally medulla agonism
- ↓brainstem response to CO2
- ↓CO2 responsiveness
- Airway effects
- Antitussive → by central opioid α agonism
- Bronchospasm → histamine release provoked by opioids
- ↓ciliary activity
- CVS
- – VE INOTROPY
- Pethidine causes marked myocardial depression
- – VE CHRONOTROPY
- Central vagal (X) excitation
- Depression of AV Nodal Conduction (exception pethidine → tachycardia)
- ↓SVR
- Histamine release → VD
- – VE INOTROPY
- GU
- Urinary retention
- Ureteric colic
- Uterine contraction
∴ opioids cause SMOOTH MUSCLE CONTRACTION
NB: Opioids can worsen pain of ureteric colic ∴ pethidine is preferred
- GI
- N&V → stimulation of µ receptors on CTZ floor 4th ventricle
- ↑smooth m. tone → delayed gastric emptying, reflux, constipation, biliary colic
- Immunological
- Histamine release (M > Pethidine > F)
NB: histamine release is not opioid R mediated & true allergy to opioids is uncommon
- MSK
- Pruritus → due to central effects & some histamine release
- Usually post neuraxial administration
- Intrathecal opioids cause pruritus by segmental excitation within SC
- Muscle rigidity → esp thorax/abdomen
- Due to excitation
- Especially with high dose opioid
- Pruritus → due to central effects & some histamine release